The Taffix (Powder) Spray Device is indicated for use against viruses within the nasal cavity.
How It Works
Taffix’s creates a hostile microenvironment in the nose where many airborne viruses can’t survive.
Protect the nose
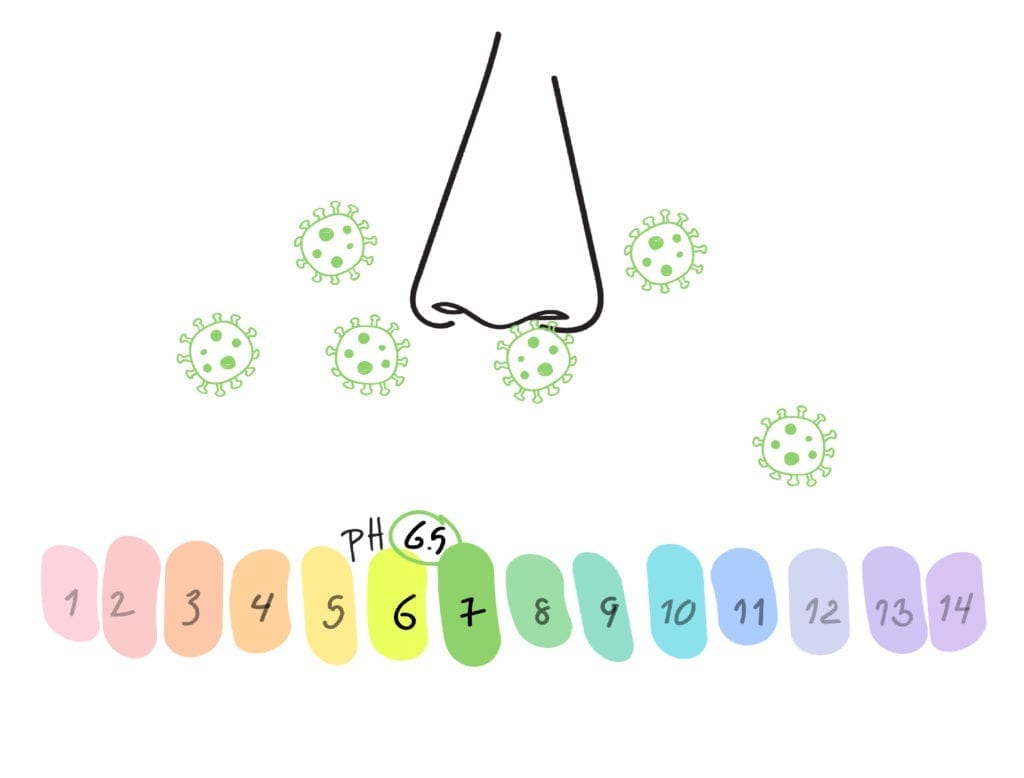
Your nose is the primary route of infection for all airborne viruses. It has a pH of 6.5 which viruses just love.
Risk reduction
Taffix creates an ultra-thin acidic gel layer that reduces the risk of virus infection in the nasal cavity

Hostile microenvironment
The mechanical protection of the acidic gel prevents viruses from entering the nasal cells and reducing the risk of infection.
Taffix Blog
How Contagious Are Respiratory Viruses in Crowded Places?
Read more
In places like airport lounges and busy offices, infection prevention is challenging. Aerosols containing germs and viruses like flu, SARS, H1N1, and rhinoviruses can remain infective for hours. Up to 48 hours in crowded rooms and busy areas, to be precise. Often, the infection rate for airborne...
The best way to prevent respiratory contagion in crowded places
Read more
Airborne viruses have always been among us. But lately, as we came to fear the unexpected, more people want to find out ways to prevent virall infection. Especially in crowded places. Viral particles move in a predictable way. But as the environmental factors change, predicting…
Practical tips for staying safe during a pandemic
Read more
During these uncertain times, it’s essential for each and everyone of us to do our best and stay protected, and do our part in stopping the spread of the virus.The good news is that with a few simple rituals, we can greatly reduce the risk...